Home > ULTRAVIOLET

|
|
||||
ULTRAVIOLET

Ultraviolet light is part of the electromagnetic spectrum which covers gamma rays through to radio waves, and radiation is waves of the electromagnetic field. The term ultraviolet is derived from the location of the specific wavelengths in the spectrum, just beyond the violet. While UV is only a small part of the electromagnetic spectrum there are many applications for its use.
Ultraviolet (UV) radiation is a form of energy transfer from one body to another with no material substance in the intervening space.
Electromagnetic waves are nature’s method of transmitting energy across space. The sun, with its enormous store of internal energy, is a primary source of radiation most familiar to us. The family of X-rays, gamma rays, UV, and infrared each share common characteristics: one they travel in straight lines, and two, can be diffracted, refracted, and polarised. Certain materials can enhance absorption, reflection and transmission of these waves.
Every wave has (3) characteristics common to all: velocity of propagation (speed); frequency; and wavelength. For example, most of us are familiar with the phenomenon of light entering a prism. When white light enters a prism, the result is a separation of the wave bands. In other words all of the colours of spectrum beaming out of the prism are a result of a decrease in speed. Frequency remains constant for a given radiation.
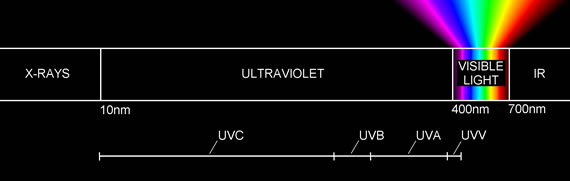
The ultraviolet light is broken into spectral ranges, UV-A 400 – 315nm, UV-B 315 – 280 nm and UV- C being 280 – 100nm,
Primarily the lower pressure mercury lamps operate in the UV-C band while the medium pressure mercury lamps operate across the bands with high intensity peaks in the UV-A and visible regions of the spectrum being 400 – 700nm
Ultraviolet (UV) radiation is a form of energy transfer from one body to another with no material substance in the intervening space.
Electromagnetic waves are nature’s method of transmitting energy across space. The sun, with its enormous store of internal energy, is a primary source of radiation most familiar to us. The family of X-rays, gamma rays, UV, and infrared each share common characteristics: one they travel in straight lines, and two, can be diffracted, refracted, and polarised. Certain materials can enhance absorption, reflection and transmission of these waves.
Every wave has (3) characteristics common to all: velocity of propagation (speed); frequency; and wavelength. For example, most of us are familiar with the phenomenon of light entering a prism. When white light enters a prism, the result is a separation of the wave bands. In other words all of the colours of spectrum beaming out of the prism are a result of a decrease in speed. Frequency remains constant for a given radiation.
The ultraviolet light is broken into spectral ranges, UV-A 400 – 315nm, UV-B 315 – 280 nm and UV- C being 280 – 100nm,
Primarily the lower pressure mercury lamps operate in the UV-C band while the medium pressure mercury lamps operate across the bands with high intensity peaks in the UV-A and visible regions of the spectrum being 400 – 700nm
APPLICATIONS
Electronics, Graphic Arts, Label Printing, CD-DVD replication, Bottle, cup and Plastic Decorating, Wood Finishing, UV-coaters and Digital Printing.
4 items




